Contents
Abstract
Rheumatoid arthritis is the most commonly diagnosed systemic inflammatory arthritis. Women, smokers, and those with a family history of the disease are most often affected. Criteria for diagnosis include having at least one joint with definite swelling that is not explained by another disease. The likelihood of a rheumatoid arthritis diagnosis increases with the number of small joints involved. In a patient with inflammatory arthritis, the presence of a rheumatoid factor or anti-citrullinated protein antibody, or elevated C-reactive protein level or erythrocyte sedimentation rate suggests a diagnosis of rheumatoid arthritis. Initial laboratory evaluation should also include complete blood count with dif- ferential and assessment of renal and hepatic function. Patients taking biologic agents should be tested for hepatitis B, hepatitis C, and tuberculosis. Earlier diagnosis of rheumatoid arthritis allows for earlier treatment with disease-modifying antirheumatic agents. Combinations of medications are often used to control the disease. Methotrexate is typically the first-line drug for rheumatoid arthritis. Biologic agents, such as tumor necrosis factor inhibitors, are generally considered second-line agents or can be added for dual therapy. The goals of treatment include minimiza- tion of joint pain and swelling, prevention of radiographic damage and visible deformity, and continuation of work and personal activities. Joint replacement is indicated for patients with severe joint damage whose symptoms are poorly controlled by medical management. (Am Fam Physician. 2011;84(11):1245-1252. Copyright � 2011 American Academy of Family Physicians.) Rheumatoid arthritis (RA) is the most common inflammatory arthritis, with a lifetime prevalence of up to 1 percent worldwide.1 Onset can occur at any age, but peaks between 30 and 50 years.2 Disability is common and significant. In a large U.S. cohort, 35 percent of patients with RA had work disability after 10 years.3Etiology and Pathophysiology
Like many autoimmune diseases, the etiology of RA is multifactorial. Genetic susceptibility is evident in familial clustering and monozygotic twin studies, with 50 percent of RA risk attributable to genetic factors.4 Genetic associations for RA include human leukocyte antigen-DR45 and -DRB1, and a variety of alleles called the shared epitope.6,7 Genome-wide association studies have identified additional genetic signatures that increase the risk of RA and other autoimmune diseases, including STAT4 gene and CD40 locus.5 Smoking is the major environmental trigger for RA, especially in those with a genetic predisposition.8 Although infections may unmask an autoimmune response, no particular pathogen has been proven to cause RA.9 RA is characterized by inflammatory pathways that lead to proliferation of synovial cells in joints. Subsequent pannus formation may lead to underlying cartilage destruction and bony erosions. Overproduction of pro-inflammatory cytokines, including tumor necrosis factor (TNF) and interleukin-6, drives the destructive process.10Risk Factors
Older age, a family history of the disease, and female sex are associated with increased risk of RA, although the sex differential is less prominent in older patients.1 Both current and prior cigarette smoking increases the risk of RA (relative risk [RR] = 1.4, up to 2.2 for more than 40-pack-year smokers).11 Pregnancy often causes RA remission, likely because of immunologic tolerance.12 Parity may have long-lasting impact; RA is less likely to be diagnosed in parous women than in nulliparous women (RR = 0.61).13,14 Breastfeeding decreases the risk of RA (RR = 0.5 in women who breastfeed for at least 24 months), whereas early menarche�(RR = 1.3 for those with menarche at 10 years of age or younger) and very irregular menstrual periods (RR = 1.5) increase risk.14 Use of oral contraceptive pills or vitamin E does not affect RA risk.15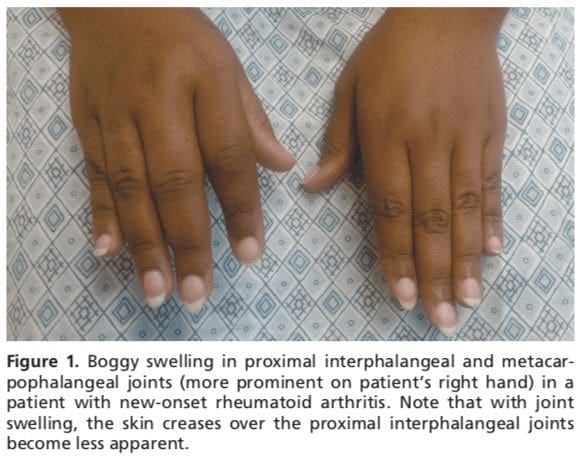
Diagnosis
Typical Presentation
Patients with RA typically present with pain and stiffness in multiple joints. The wrists, proximal interphalangeal joints, and metacarpophalangeal joints are most commonly involved. Morning stiffness lasting more than one hour suggests an inflammatory etiology. Boggy swelling due to synovitis may be visible (Figure 1), or subtle synovial thickening may be palpable on joint examination. Patients may also present with more indolent arthralgias before the onset of clinically apparent joint swelling. Systemic symptoms of fatigue, weight loss, and low-grade fever may occur with active disease.Diagnostic Criteria
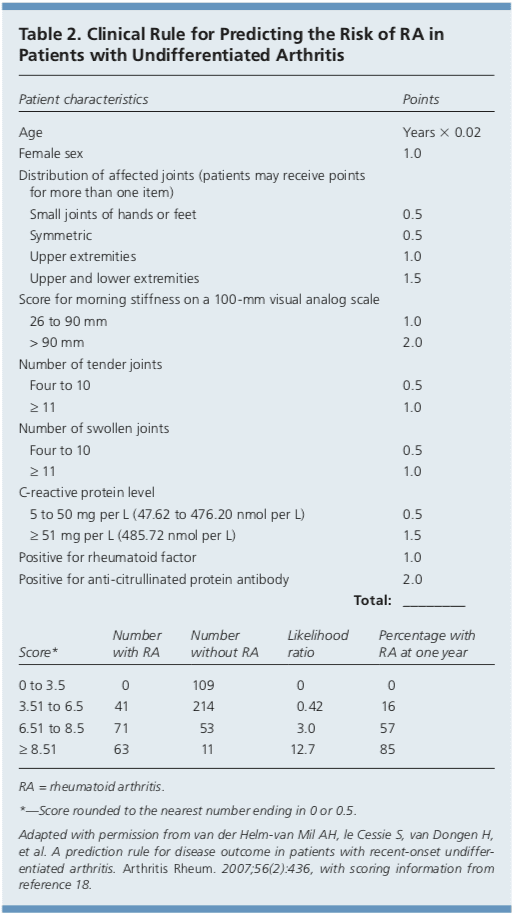
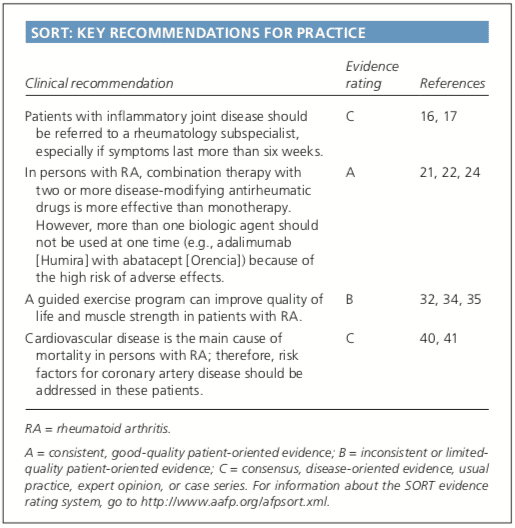
In 2010, the American College of Rheumatology and European League Against Rheumatism collaborated to create new classification criteria for RA (Table 1).16 The new criteria are an effort to diagnose RA earlier in patients who may not meet the 1987 American College of Rheumatology classification criteria. The 2010 criteria do not include presence of rheumatoid nodules or radiographic erosive changes, both of which are less likely in early RA. Symmetric arthri- tis is also not required in the 2010 criteria, allowing for early asymmetric presentation. In addition, Dutch researchers have developed and validated a clinical prediction rule for RA (Table 2).17,18 The purpose of this rule is to help identify patients with undifferentiated arthritis that is most likely to progress to RA, and to guide follow-up and referral.Diagnostic Tests
Autoimmune diseases such as RA are often characterized by the presence of autoanti- bodies. Rheumatoid factor is not specific for RA and may be present in patients with other diseases, such as hepatitis C, and in healthy older persons. Anti-citrullinated protein antibody is more specific for RA and may play a role in disease pathogenesis.6 Approxi- mately 50 to 80 percent of persons with RA have rheumatoid factor, anti-citrullinated protein antibody, or both.10 Patients with RA may have a positive antinuclear antibody test result, and the test is of prognostic impor- tance in juvenile forms of this disease.19 C-reactive protein levels and erythrocyte sedimentation rate are often increased with active RA, and these acute phase reactants are part of the new RA classification criteria.16 C-reactive protein levels and erythrocyte sedimentation rate may also be used to follow disease activity and response to medication. Baseline complete blood count with differential and assessment of renal and hepatic function are helpful because the results may influence treatment options (e.g., a patient with renal insufficiency or significant thrombocytopenia likely would not be prescribed a nonsteroidal anti-inflammatory drug [NSAID]). Mild anemia of chronic disease occurs in 33 to 60 percent of all patients with RA,20 although gastrointestinal blood loss should also be considered in patients taking corticosteroids or NSAIDs. Methotrexate is contraindicated in patients with hepatic disease, such as hepatitis C, and in patients with significant renal impairment.21 Biologic therapy, such as a TNF inhibitor, requires a negative tuberculin test or treatment for latent tuberculosis. Hepatitis B reactivation can also occur with TNF inhibitor use.22 Radiography of hands and feet should be performed to evaluate for characteristic periarticular erosive changes,�which may be indicative of a more aggressive RA subtype.10
Differential Diagnosis
Skin findings suggest systemic lupus erythematosus, systemic sclerosis, or psoriatic arthritis. Polymyalgia rheumatica should be considered in an older patient with symptoms primarily in the shoulder and hip, and the patient should be asked questions related to associated temporal arteritis. Chest radiography is helpful to evaluate for sarcoidosis as an etiology of arthritis.�Patients with inflammatory back symptoms, a history of inflammatory bowel disease, or inflammatory eye disease may have spondyloarthropathy. Persons with less than six weeks of symptoms may have a viral process, such as parvovirus. Recurrent self-limited episodes of acute joint swelling suggest crystal arthropathy, and arthrocentesis should be performed to evaluate for monosodium urate monohydrate or calcium pyrophosphate dihydrate crystals. The presence of numerous myofascial trigger points and somatic symptoms may suggest fibromyalgia, which can coexist with RA. To help guide diagnosis and determine treatment strategy, patients with inflammatory arthritis should be promptly referred to a rheumatology subspecialist.16,17
Rheumatoid arthritis, or RA, is the most common type of arthritis. RA is an autoimmune disease, caused when the immune system, the human body’s defense system, attacks its own cells and tissues, particularly the joints. Rheumatoid arthritis is frequently identified by symptoms of pain and inflammation, often affecting the small joints of the hands, wrists and feet. According to many healthcare professionals, early diagnosis and treatment of RA is essential to prevent further joint damage and decrease painful symptoms. Dr. Alex Jimenez D.C., C.C.S.T. Insight
Treatment
After RA has been diagnosed and an initial evaluation performed, treatment should begin. Recent guidelines have addressed the management of RA,21,22 but patient preference also plays an important role. There are special considerations for women of childbearing age because many medications have deleterious effects on pregnancy. Goals of therapy include minimizing joint pain and swelling, preventing deformity (such as ulnar deviation) and radiographic damage (such as erosions), maintaining quality of life (personal and work), and controlling extra-articular manifestations. Disease-modifying antirheumatic drugs (DMARDs) are the mainstay of RA therapy.DMARDs
DMARDs can be biologic or nonbiologic (Table 3).23 Biologic agents include monoclonal antibodies and recombinant receptors to block cytokines that promote the inflammatory cascade responsible for RA symptoms. Methotrexate is recommended as the first- line treatment in patients with active RA, unless contraindicated or not tolerated.21 Leflunomide (Arava) may be used as an alternative to methotrexate, although gastrointestinal adverse effects are more common. Sulfasalazine (Azulfidine) or hydroxychloroquine (Plaquenil) pro-inflammatory as monotherapy in patients with low disease�activity or without poor prognostic features (e.g., seronegative, non-erosive RA).21,22 Combination therapy with two or more DMARDs is more effective than monotherapy; however, adverse effects may also be greater.24 If RA is not well controlled with a nonbiologic DMARD, a biologic DMARD should be initiated.21,22 TNF inhibitors are the first-line biologic therapy and are the most studied of these agents. If TNF inhibitors are ineffective, additional biologic therapies can be considered. Simultaneous use of more than one biologic therapy (e.g., adalimumab [Humira] with abatacept [Orencia]) is not�recommended because of an unacceptable rate of adverse effects.21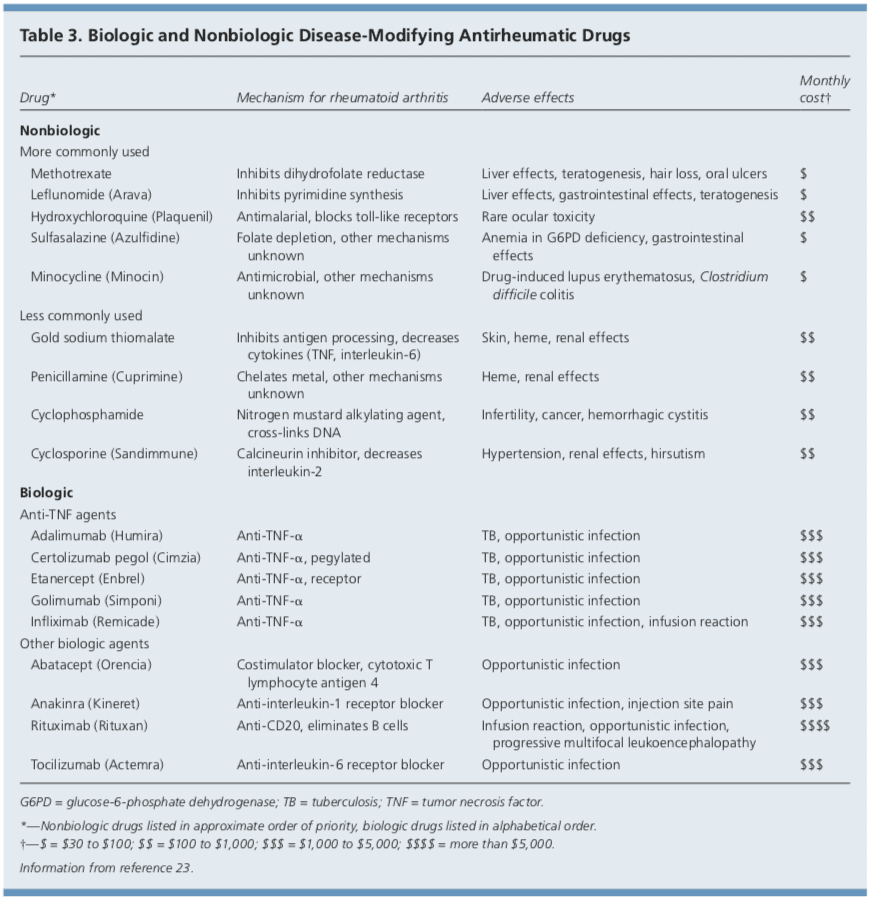
NSAIDs and Corticosteroids
Drug therapy for RA may involve NSAIDs and oral, intramuscular, or intra-articular corticosteroids for controlling pain and inflammation. Ideally, NSAIDs and corticosteroids are used only for short-term management. DMARDs are the preferred therapy.21,22Complementary Therapies
Dietary interventions, including vegetarian and Mediterranean diets, have been�studied in the treatment of RA without convincing evidence of benefit.25,26 Despite some favorable outcomes, there is a lack of evidence for the effectiveness of acupuncture in placebo-controlled trials of patients with RA.27,28 In addition, thermotherapy and therapeutic ultrasound for RA have not been studied adequately.29,30 A Cochrane review of herbal treatments for RA concluded that gamma-linolenic acid (from evening primrose or black currant seed oil) and Tripterygium wilfordii (thunder god vine) have potential benefits.31 It is important to inform patients that serious adverse effects have been reported with use of herbal therapy.31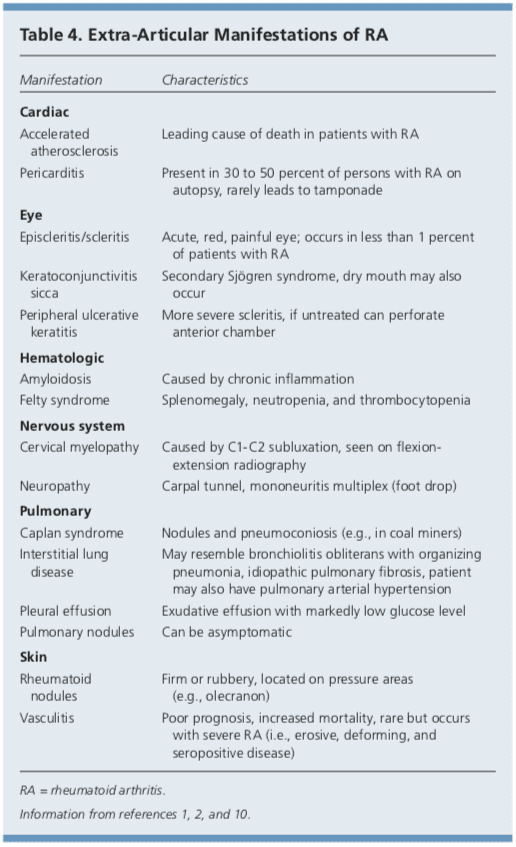
Exercise and Physical Therapy
Results of randomized controlled trials sup- port physical exercise to improve quality of life and muscle strength in patients with RA.32,33 Exercise training programs have not been shown to have deleterious effects on RA disease activity, pain scores, or radiographic joint damage.34 Tai chi has been shown to improve ankle range of motion in persons with RA, although randomized trials are limited.35 Randomized controlled trials of Iyengar yoga in young adults with RA are underway.36Duration of Treatment
Remission is obtainable in 10 to 50 percent of patients with RA, depending on how remission is defined and the intensity of therapy.10 Remission is more likely in males, nonsmokers, persons younger than 40 years, and in those with late-onset disease (patients older than 65 years), with shorter duration of disease, with milder disease activity, without elevated acute phase reactants, and without positive rheumatoid factor or anti-citrullinated protein antibody findings.37 After the disease is controlled, medication dosages may be cautiously decreased to the minimum amount necessary. Patients will require frequent monitoring to ensure stable symptoms, and prompt increase in medication is recommended with disease flare-ups.22Joint Replacement
Joint replacement is indicated when there is severe joint damage and unsatisfactory control of symptoms with medical management. Long-term outcomes are support, with only 4 to 13 percent of large joint replacements requiring revision within 10 years.38 The hip and knee are the most commonly replaced joints.Long-Term Monitoring
Although RA is considered a disease of the joints, it is also a systemic disease capable of involving multiple organ systems. Extra-articular manifestations of RA are included in Table 4.1,2,10 Patients with RA have a twofold increased risk of lymphoma, which is thought to be caused by the underlying inflammatory�process, and not a consequence of medical treatment.39 Patients with RA are also at an increased risk of coronary artery disease, and physicians should work with patients to modify risk factors, such as smoking, high blood pressure, and high cholesterol.40,41 Class III or IV congestive heart failure (CHF) is a contraindication for using TNF inhibitors, which can worsen CHF outcomes.21 In patients with RA and malignancy, caution is needed with continued use of DMARDs, especially TNF inhibitors. Biologic DMARDs, methotrexate, and leflunomide should not be initiated in patients with active herpes zoster, significant fungal infection, or bacterial infection requiring antibiotics.21 Complications of RA and its treatments are listed in Table 5.1,2,10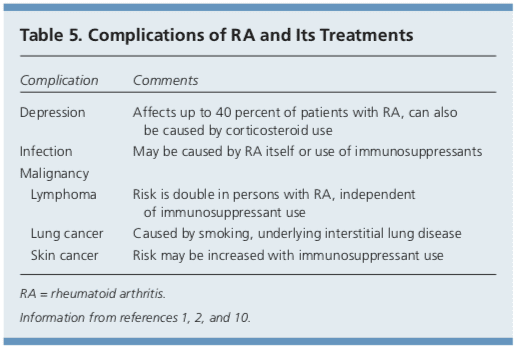
Prognosis
Patients with RA live three to 12 years less than the general population.40 Increased mortality in these patients is mainly due to accelerated cardiovascular disease, especially in those with high disease activity and chronic inflammation. The relatively new biologic therapies may reverse progression of atherosclerosis and extend life in those with RA.41 Data Sources: A PubMed search was completed in Clinical Queries using the key terms rheumatoid arthritis, extra-articular manifestations, and disease-modifying antirheumatic agents. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. Also searched were the Agency for Healthcare Research and Quality evidence reports, Clinical Evidence, the Cochrane database, Essential Evidence, and UpToDate. Search date: September 20, 2010. Author disclosure: No relevant financial affiliations to disclose. In conclusion, rheumatoid arthritis is a chronic, autoimmune disease which causes painful symptoms, such as pain and discomfort, inflammation and swelling of the joints, among others. The joint damage characterized as RA is symmetrical, meaning it generally affects both sides of the body. Early�diagnosis is essential for treatment of RA. The scope of our information is limited to chiropractic and spinal health issues. To discuss the subject matter, please feel free to ask Dr. Jimenez or contact us at�915-850-0900�. Curated by Dr. Alex Jimenez
Additional Topic Discussion: Relieving Knee Pain without Surgery
Knee pain is a well-known symptom which can occur due to a variety of knee injuries and/or conditions, including�sports injuries. The knee is one of the most complex joints in the human body as it is made-up of the intersection of four bones, four ligaments, various tendons, two menisci, and cartilage. According to the American Academy of Family Physicians, the most common causes of knee pain include patellar subluxation, patellar tendinitis or jumper’s knee, and Osgood-Schlatter disease. Although knee pain is most likely to occur in people over 60 years old, knee pain can also occur in children and adolescents. Knee pain can be treated at home following the RICE methods, however, severe knee injuries may require immediate medical attention, including chiropractic care.
EXTRA EXTRA | IMPORTANT TOPIC: El Paso, TX Chiropractor Recommended
Blank
References
1. Etiology and pathogenesis of rheumatoid arthritis. In: Firestein GS, Kelley WN, eds. Kelley�s Textbook of Rheu- matology. 8th ed. Philadelphia, Pa.: Saunders/Elsevier; 2009:1035-1086.
2. Bathon J, Tehlirian C. Rheumatoid arthritis clinical and
laboratory manifestations. In: Klippel JH, Stone JH, Crofford LJ, et al., eds. Primer on the Rheumatic Dis- eases. 13th ed. New York, NY: Springer; 2008:114-121.
3. Allaire S, Wolfe F, Niu J, et al. Current risk factors for work disability associated with rheumatoid arthritis. Arthritis Rheum. 2009;61(3):321-328.
4. MacGregor AJ, Snieder H, Rigby AS, et al. Characteriz- ing the quantitative genetic contribution to rheumatoid arthritis using data from twins. Arthritis Rheum. 2000; 43(1):30-37.
5. Orozco G, Barton A. Update on the genetic risk fac- tors for rheumatoid arthritis. Expert Rev Clin Immunol. 2010;6(1):61-75.
6. Balsa A, Cabezo?n A, Orozco G, et al. Influence of HLA DRB1 alleles in the susceptibility of rheumatoid arthritis and the regulation of antibodies against citrullinated proteins and rheumatoid factor. Arthritis Res Ther. 2010;12(2):R62.
7. McClure A, Lunt M, Eyre S, et al. Investigating the via- bility of genetic screening/testing for RA susceptibility using combinations of five confirmed risk loci. Rheuma- tology (Oxford). 2009;48(11):1369-1374.
8. Bang SY, Lee KH, Cho SK, et al. Smoking increases rheu- matoid arthritis susceptibility in individuals carrying the HLA-DRB1 shared epitope, regardless of rheumatoid factor or anti-cyclic citrullinated peptide antibody sta- tus. Arthritis Rheum. 2010;62(2):369-377.
9. Wilder RL, Crofford LJ. Do infectious agents cause rheu- matoid arthritis? Clin Orthop Relat Res. 1991;(265): 36-41.
10. Scott DL, Wolfe F, Huizinga TW. Rheumatoid arthritis. Lancet. 2010;376(9746):1094-1108.
11. Costenbader KH, Feskanich D, Mandl LA, et al. Smoking intensity, duration, and cessation, and the risk of rheu- matoid arthritis in women. Am J Med. 2006;119(6): 503.e1-e9.
12. Kaaja RJ, Greer IA. Manifestations of chronic disease during pregnancy. JAMA. 2005;294(21):2751-2757.
13. Guthrie KA, Dugowson CE, Voigt LF, et al. Does preg-
nancy provide vaccine-like protection against rheuma-
toid arthritis? Arthritis Rheum. 2010;62(7):1842-1848.
14. Karlson EW, Mandl LA, Hankinson SE, et al. Do breast- feeding and other reproductive factors influence future risk of rheumatoid arthritis? Results from the Nurses� Health Study. Arthritis Rheum. 2004;50(11):3458-3467.
15. Karlson EW, Shadick NA, Cook NR, et al. Vitamin E in the primary prevention of rheumatoid arthritis: the Women�s Health Study. Arthritis Rheum. 2008;59(11):
1589-1595.
16. Aletaha D, Neogi T, Silman AJ, et al. 2010 rheumatoid
arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative [published correction appears in Ann Rheum Dis. 2010;69(10):1892]. Ann Rheum Dis. 2010;69(9):1580-1588.
17. van der Helm-van Mil AH, le Cessie S, van Dongen H, et al. A prediction rule for disease outcome in patients with recent-onset undifferentiated arthritis. Arthritis Rheum. 2007;56(2):433-440.
18. Mochan E, Ebell MH. Predicting rheumatoid arthritis risk in adults with undifferentiated arthritis. Am Fam Physi- cian. 2008;77(10):1451-1453.
19. Ravelli A, Felici E, Magni-Manzoni S, et al. Patients with antinuclear antibody-positive juvenile idiopathic arthri- tis constitute a homogeneous subgroup irrespective of the course of joint disease. Arthritis Rheum. 2005; 52(3):826-832.
20. Wilson A, Yu HT, Goodnough LT, et al. Prevalence and outcomes of anemia in rheumatoid arthritis. Am J Med. 2004;116(suppl 7A):50S-57S.
21. Saag KG, Teng GG, Patkar NM, et al. American College of Rheumatology 2008 recommendations for the use of nonbiologic and biologic disease-modifying antirheu- matic drugs in rheumatoid arthritis. Arthritis Rheum. 2008;59(6):762-784.
22. Deighton C, O�Mahony R, Tosh J, et al.; Guideline Devel- opment Group. Management of rheumatoid arthritis: summary of NICE guidance. BMJ. 2009;338:b702.
23. AHRQ. Choosing medications for rheumatoid arthritis. April 9, 2008. http://www.effectivehealthcare.ahrq.gov/ ehc/products/14/85/RheumArthritisClinicianGuide.pdf. Accessed June 23, 2011.
24. Choy EH, Smith C, Dore? CJ, et al. A meta-analysis of the efficacy and toxicity of combining disease-modify- ing anti-rheumatic drugs in rheumatoid arthritis based on patient withdrawal. Rheumatology (Oxford). 2005; 4 4 (11) :1414 -1421.
25. Smedslund G, Byfuglien MG, Olsen SU, et al. Effective- ness and safety of dietary interventions for rheumatoid arthritis. J Am Diet Assoc. 2010;110(5):727-735.
26. Hagen KB, Byfuglien MG, Falzon L, et al. Dietary inter- ventions for rheumatoid arthritis. Cochrane Database Syst Rev. 2009;21(1):CD006400.
27. Wang C, de Pablo P, Chen X, et al. Acupuncture for pain relief in patients with rheumatoid arthritis: a systematic review. Arthritis Rheum. 2008;59(9):1249-1256.
28. Kelly RB. Acupuncture for pain. Am Fam Physician. 2009;80(5):481-484.
29. Robinson V, Brosseau L, Casimiro L, et al. Thermother- apy for treating rheumatoid arthritis. Cochrane Data- base Syst Rev. 2002;2(2):CD002826.
30. Casimiro L, Brosseau L, Robinson V, et al. Therapeutic ultrasound for the treatment of rheumatoid arthritis. Cochrane Database Syst Rev. 2002;3(3):CD003787.
31. Cameron M, Gagnier JJ, Chrubasik S. Herbal therapy for treating rheumatoid arthritis. Cochrane Database Syst Rev. 2011;(2):CD002948.
32. Brodin N, Eurenius E, Jensen I, et al. Coaching patients with early rheumatoid arthritis to healthy physical activ- ity. Arthritis Rheum. 2008;59(3):325-331.
33. Baillet A, Payraud E, Niderprim VA, et al. A dynamic exercise programme to improve patients� disability in rheumatoid arthritis: a prospective randomized con- trolled trial. Rheumatology (Oxford). 2009;48(4): 410-415.
34. Hurkmans E, van der Giesen FJ, Vliet Vlieland TP, et al. Dynamic Exercise programs (aerobic capacity and/or mus- cle strength training) in patients with rheumatoid arthri- tis. Cochrane Database Syst Rev. 2009;(4):CD006853.
35. Han A, Robinson V, Judd M, et al. Tai chi for treat- ing rheumatoid arthritis. Cochrane Database Syst Rev. 2004;(3):CD004849.
36. Evans S, Cousins L, Tsao JC, et al. A randomized con- trolled trial examining Iyengar yoga for young adults with rheumatoid arthritis. Trials. 2011;12:19.
37. Katchamart W, Johnson S, Lin HJ, et al. Predictors for remis- sion in rheumatoid arthritis patients: a systematic review. Arthritis Care Res (Hoboken). 2010;62(8):1128-1143.
38. Wolfe F, Zwillich SH. The long-term outcomes of rheu- matoid arthritis: a 23-year prospective, longitudinal study of total joint replacement and its predictors in 1,600 patients with rheumatoid arthritis. Arthritis Rheum. 1998;41(6):1072-1082.
39. Baecklund E, Iliadou A, Askling J, et al. Association of chronic inflammation, not its treatment, with increased lymphoma risk in rheumatoid arthritis. Arthritis Rheum. 2006;54(3):692-701.
40. Friedewald VE, Ganz P, Kremer JM, et al. AJC editor�s consensus: rheumatoid arthritis and atherosclerotic cardiovascular disease. Am J Cardiol. 2010;106(3): 442-447.
41. Atzeni F, Turiel M, Caporali R, et al. The effect of phar- macological therapy on the cardiovascular system of patients with systemic rheumatic diseases. Autoimmun Rev. 2010;9(12):835-839.
Close Accordion
General Disclaimer, Licenses and Board Certifications *
Professional Scope of Practice *
The information herein on "Diagnosis and Management of Rheumatoid Arthritis" is not intended to replace a one-on-one relationship with a qualified health care professional or licensed physician and is not medical advice. We encourage you to make healthcare decisions based on your research and partnership with a qualified healthcare professional.
Blog Information & Scope Discussions
Welcome to El Paso's Premier Wellness and Injury Care Clinic & Wellness Blog, where Dr. Alex Jimenez, DC, FNP-C, a Multi-State board-certified Family Practice Nurse Practitioner (FNP-BC) and Chiropractor (DC), presents insights on how our multidisciplinary team is dedicated to holistic healing and personalized care. Our practice aligns with evidence-based treatment protocols inspired by integrative medicine principles, similar to those on this site and on our family practice-based chiromed.com site, focusing on naturally restoring health for patients of all ages.
Our areas of multidisciplinary practice include Wellness & Nutrition, Chronic Pain, Personal Injury, Auto Accident Care, Work Injuries, Back Injury, Low Back Pain, Neck Pain, Migraine Headaches, Sports Injuries, Severe Sciatica, Scoliosis, Complex Herniated Discs, Fibromyalgia, Chronic Pain, Complex Injuries, Stress Management, Functional Medicine Treatments, and in-scope care protocols.
Our information scope is multidisciplinary, focusing on musculoskeletal and physical medicine; wellness; contributing etiological viscerosomatic disturbances within clinical presentations; associated somato-visceral reflex clinical dynamics; subluxation complexes; sensitive health issues; and functional medicine articles, topics, and discussions.
We provide and present clinical collaboration with specialists from various disciplines. Each specialist is governed by their professional scope of practice and licensure jurisdiction. We use functional health & wellness protocols to treat and support care for musculoskeletal injuries or disorders.
Our videos, posts, topics, and insights address clinical matters and issues that directly or indirectly relate to our clinical scope of practice.
Our office has made a reasonable effort to provide supportive citations and has identified relevant research studies that support our posts. We provide copies of supporting research studies upon request to regulatory boards and the public.
We understand that we cover matters that require an additional explanation of how they may assist in a particular care plan or treatment protocol; therefore, to discuss the subject matter above further, please feel free to ask Dr. Alex Jimenez, DC, APRN, FNP-BC, or contact us at 915-850-0900.
We are here to help you and your family.
Blessings
Dr. Alex Jimenez, DC, MSACP, APRN, FNP-BC*, CCST, IFMCP, CFMP, ATN
email: [email protected]
Multidisciplinary Licensing & Board Certifications:
Licensed as a Doctor of Chiropractic (DC) in Texas & New Mexico*
Texas DC License #: TX5807, Verified: TX5807
New Mexico DC License #: NM-DC2182, Verified: NM-DC2182
Multi-State Advanced Practice Registered Nurse (APRN*) in Texas & Multi-States
Multi-state Compact APRN License by Endorsement (42 States)
Texas APRN License #: 1191402, Verified: 1191402 *
Florida APRN License #: 11043890, Verified: APRN11043890 *
Colorado License #: C-APN.0105610-C-NP, Verified: C-APN.0105610-C-NP
New York License #: N25929, Verified N25929
License Verification Link: Nursys License Verifier
* Prescriptive Authority Authorized
ANCC FNP-BC: Board Certified Nurse Practitioner*
Compact Status: Multi-State License: Authorized to Practice in 40 States*
Graduate with Honors: ICHS: MSN-FNP (Family Nurse Practitioner Program)
Degree Granted. Master's in Family Practice MSN Diploma (Cum Laude)
Dr. Alex Jimenez, DC, APRN, FNP-BC*, CFMP, IFMCP, ATN, CCST
(Board Certified: Family Practice Nurse Practitioner—Multistate)*
(Licensed Nurse Practitioner & Chiropractor - Multistate)*
Clinical Director
Digital Business Card
Dr. Maria Cardenas, MD
(Board Certified: Internal Medicine)
(Licensed Medical Doctor)
Medical Director, Clinical Director & Collaborative Physician
NPI # 1164426749
MD License #: J2933
Licenses and Board Certifications:
MD: Medical Doctor
DC: Doctor of Chiropractic
APRNP: Advanced Practice Registered Nurse
FNP-BC: Family Practice Specialization (Multi-State Board Certified)
RN: Registered Nurse (Multi-State Compact License)
CFMP: Certified Functional Medicine Provider
MSN-FNP: Master of Science in Family Practice Medicine
MSACP: Master of Science in Advanced Clinical Practice
IFMCP: Institute of Functional Medicine
CCST: Certified Chiropractic Spinal Trauma
ATN: Advanced Translational Neutrogenomics
Memberships & Associations:
TCA: Texas Chiropractic Association: Member ID: 104311
AANP: American Association of Nurse Practitioners: Member ID: 2198960
ANA: American Nurse Association: Member ID: 06458222 (District TX01)
TNA: Texas Nurse Association: Member ID: 06458222
NPI: 1205907805
| Primary Taxonomy | Selected Taxonomy | State | License Number |
|---|---|---|---|
| No | 111N00000X - Chiropractor | NM | DC2182 |
| Yes | 111N00000X - Chiropractor | TX | DC5807 |
| Yes | 363LF0000X - Nurse Practitioner - Family | TX | 1191402 |
| Yes | 363LF0000X - Nurse Practitioner - Family | FL | 11043890 |
| Yes | 363LF0000X - Nurse Practitioner - Family | CO | C-APN.0105610-C-NP |
| Yes | 363LF0000X - Nurse Practitioner - Family | NY | N25929 |
Dr. Alex Jimenez, DC, APRN, FNP-BC*, CFMP, IFMCP, ATN, CCST
(Board Certified: Family Practice Nurse Practitioner—Multistate)*
(Licensed Nurse Practitioner & Chiropractor - Multistate)*
Clinical Director
Digital Business Card
Dr. Maria Cardenas, MD
(Board Certified: Internal Medicine)*
(Licensed Medical Doctor)*
Medical Director, Clinical Director & Collaborative Physician
NPI # 1164426749
MD License #: J2933